Biotech
Celltrion looks beyond biosimilars to develop its own mRNA drugs
The Korean firm also aims to secure various drug development platforms such as ADC to grow its business
By Nov 16, 2021 (Gmt+09:00)
4
Min read
Most Read
LG Chem to sell water filter business to Glenwood PE for $692 million


KT&G eyes overseas M&A after rejecting activist fund's offer


Kyobo Life poised to buy Japan’s SBI Group-owned savings bank


StockX in merger talks with Naver’s online reseller Kream


Meritz backs half of ex-manager’s $210 mn hedge fund


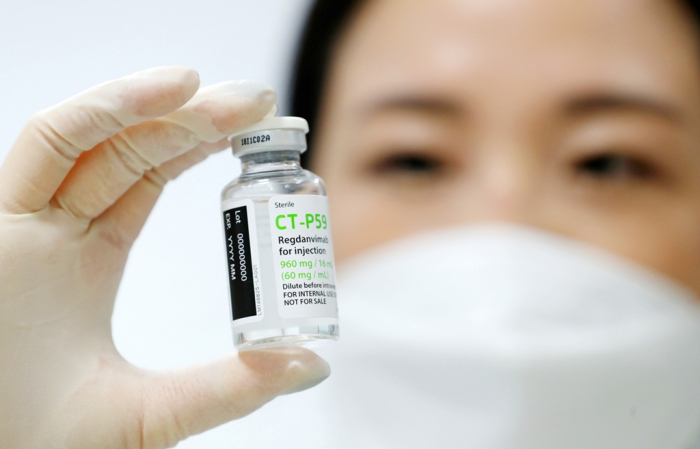
South Korea’s Celltrion Inc. has long been a biosimilar player, producing replicas of original drugs, a growing market in the pandemic era.
Earlier this week, Celltrion went out of its way to become the world’s first biosimilar maker to win approval from the European Commission for the sale in Europe of a monoclonal antibody COVID-19 treatment.
The approval by the European Medicines Agency (EMA) has also made Celltrion Korea’s first biosimilar firm to enter the European market with its own drug development technology.
With the authorization of its product, Regkirona, the company is now looking to develop its own messenger ribonucleic acid (mRNA) vaccines and secure drug platform technology to go beyond its mainstay biosimilar products.
“The authorization proves our technological prowess to develop our own new drugs, not just biosimilars. We expect the breakthrough to pave the way for us to further expand into the global biopharmaceutical market,” said Kee Woo-sung, Celltrion’s chief executive and vice chairman.
In February, Rekirona won the Korean drug safety agency’s approval, becoming the first locally made treatment for COVID-19, and is seeking approval from other countries, including the US.
Company officials said the company is now setting its sights on new drug development with its own technology as well as consolidating its position as a global biosimilar player.

WORKING ON mRNA VACCINE
In August, Celltrion said it tied up with TriLink BioTechnologies Inc., a US life sciences company, to develop a next-generation mRNA vaccine platform that can be applied to coronavirus variants.
The move came as the Korean government plans to have local drug makers and biotech companies develop their own mRNA vaccines by next year.
The mRNA vaccine, employed by Pfizer Inc. and Moderna Inc., is a type that uses a copy of a natural chemical called messenger RNA to produce an immune response.
Messenger RNA has been used to treat hepatosis and rare diseases, but given its relatively short development time, the RNA molecule is now used not just for vaccines but for the treatment of cancer and other illnesses.
Biopharmaceutical companies have been active in forging partnerships with and pursuing acquisitions of smaller competitors specialized in mRNA vaccine development.
Some other Korean companies working on mRNA vaccines include Samsung Biologics Co., Gene One Life Science Inc. and Quratis Co.
“We are considering applying the mRNA technology to develop not just COVID-19 vaccines but also anticancer medicines and vaccines for other diseases such as shingles,” said a Celltrion official.

ADC-BASED DRUGS
Celltrion is also striving to secure new drug development platforms such as antibody-drug conjugate (ADC), a next-generation technology designed to specifically target cancer cells.
The company said in June it joined hands with Mirae Asset Group to invest a combined $47 million in Iksuda Therapeutics, a UK-based ADC developer.
Celltrion expects the investment to expand its cancer drug pipeline beyond blood cancer treatment Truxima and breast cancer medication Herzuma. Iksuda has four ADC-based biopharmaceutical drugs in preclinical development, including for B-cell lymphomas.
The Korean biosimilar maker entered the ADC space in 2019 when it inked a deal with iProgen Biotech, an Indian biotech startup, to develop ADCs.
Celltrion said that ADC-based treatments qualify for fast-track drug development because most of them are designated for rare diseases or otherwise innovative.
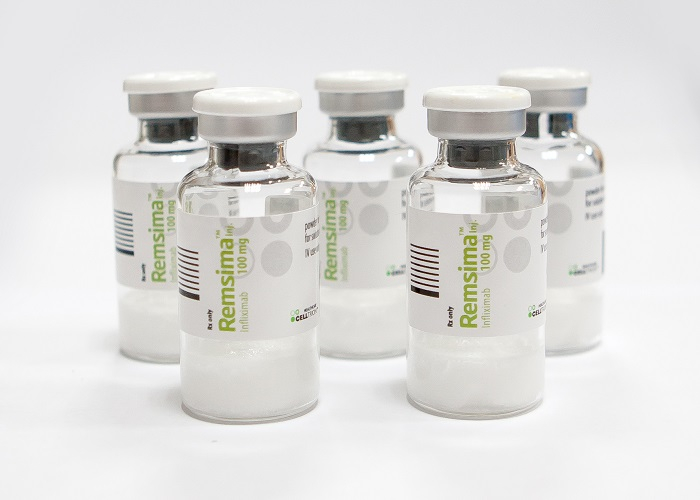
BIOSIMILARS, A CASH COW
The company, which controls more than half the biosimilar market of Janssen's Remicade in Europe with its monoclonal antibody biosimilar Remsima, said it will continue to expand its biosimilar business to secure funds needed to start new promising businesses.
Celltrion has so far launched four biosimilar products globally and has five drugs in the pipeline, with an aim to launch at least one every year until 2025.
It is seeking approval from the authorities in Korea, the US and Europe for its anticancer biosimilar candidate Avastin (Bevacizumab). Four other pipeline products are currently in the final phase 3 clinical trials.
Bevacizumab, sold under the brand name Avastin, is a medication used to treat a number of types of cancers, including colorectal, lung and kidney cancer.
Celltrion, which unveiled the world’s first subcutaneous (SC)-injection type of Remsima, a biosimilar version of Janssen’s Remicade that is also a brand name of infliximab, said last year it is working to develop a pill version of infliximab.
Write to Jae-young Han and Ju-Hyun Lee at jyhan@hankyung.com
In-Soo Nam edited this article.
More to Read
-
 COVID-19 treatmentCelltrion wins COVID-19 treatment approval from Europe
COVID-19 treatmentCelltrion wins COVID-19 treatment approval from EuropeNov 15, 2021 (Gmt+09:00)
4 Min read -
 COVID-19 vaccinesWar for bio talent intensifies as S.Korea works on mRNA vaccines
COVID-19 vaccinesWar for bio talent intensifies as S.Korea works on mRNA vaccinesAug 05, 2021 (Gmt+09:00)
3 Min read -
 COVID-19 vaccineKorea’s pharma consortium to develop mRNA vaccines by 2022
COVID-19 vaccineKorea’s pharma consortium to develop mRNA vaccines by 2022Jun 30, 2021 (Gmt+09:00)
2 Min read -
 COVID-19 treatmentCelltrion says COVID-19 antibody kills virus within 5 days; shares surge
COVID-19 treatmentCelltrion says COVID-19 antibody kills virus within 5 days; shares surgeNov 11, 2020 (Gmt+09:00)
2 Min read -
 Celltrion group tipped to exceed 1 trillion won in profit; first among bio firms
Celltrion group tipped to exceed 1 trillion won in profit; first among bio firmsSep 15, 2020 (Gmt+09:00)
2 Min read
Comment 0
LOG IN


