Covid-19 treatment
Indonesia grants emergency use approval for Celltrion’s COVID-19 treatment
Regkirona is likely to get European Medicines Agency approval by October this year as well
By Jul 20, 2021 (Gmt+09:00)
1
Min read
Most Read
LG Chem to sell water filter business to Glenwood PE for $692 million


Kyobo Life poised to buy Japan’s SBI Group-owned savings bank


KT&G eyes overseas M&A after rejecting activist fund's offer


StockX in merger talks with Naver’s online reseller Kream


Mirae Asset to be named Korea Post’s core real estate fund operator


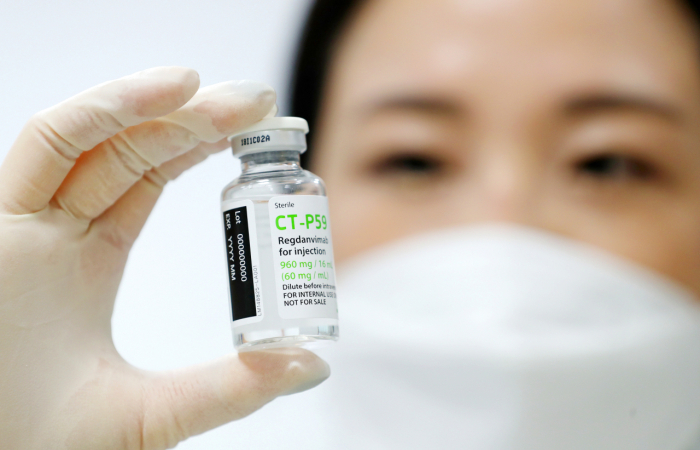
South Korean drugmaker Celltrion Inc. has received the Indonesian government’s permission for restricted use of its COVID-19 treatment Regkirona.
Celltrion said on July 20 that Indonesia’s National Agency of Drug and Food Control (Badan Pengawas Obat dan Makanan) has granted emergency use approval for its treatment.
The permission marks Regkirona’s first approval by an overseas health authority. The scope of Regkirona use is wider in Indonesia than in South Korea, where Celltrion’s treatment can be used only on high-risk patients or those older than 60.
In contrast, Indonesia has allowed Regkirona to be used on patients with mild symptoms or with underlying conditions among patients older than 50. For those under 50, Regkirona can be used on patients with severe symptoms only.

“We expect sales of Regkirona to be much higher in Indonesia where the treatment can be applied to patients with milder symptoms as well,” said a Celltrion official.
Celltrion said that it confirmed the efficacy of Regkirona in treating the delta variant through animal tests earlier this month. The delta variant is spreading fast in Indonesia, driving the average number of daily confirmed cases from less than 20,000 in mid-June to more than 50,000 last week. The total number of confirmed cases in the country was about 3 million as of July 19.
Celltrion is awaiting the approval of Regkirona in a number of other countries and regions.
Regkirona is currently under rolling review by the European Medicines Agency (EMA). A rolling review is a regulatory tool that EMA uses to speed up the assessment of a promising medicine during a public health emergency. Industry sources say that Regkirona is likely to get EMA approval by October of this year.
In the US, Celltrion is having a preliminary discussion with the Food and Drug Administration (FDA) regarding the regulatory filing.
The company added that it has already filed for Regkirona’s approval in a number of countries in the Middle East, including Iraq, Jordan, Morocco, Saudi Arabia and the UAE.
Write to Woo-sub Kim at duter@hankyung.com
Daniel Cho edited this article.
More to Read
-
 Business & PoliticsTrump Jr. meets Korean business chiefs in back-to-back sessions
Business & PoliticsTrump Jr. meets Korean business chiefs in back-to-back sessionsApr 30, 2025 (Gmt+09:00)
-
 Korean chipmakersSamsung in talks to supply customized HBM4 to Nvidia, Broadcom, Google
Korean chipmakersSamsung in talks to supply customized HBM4 to Nvidia, Broadcom, GoogleApr 30, 2025 (Gmt+09:00)
-
 EnergyLS Cable breaks ground on $681 mn underwater cable plant in Chesapeake
EnergyLS Cable breaks ground on $681 mn underwater cable plant in ChesapeakeApr 29, 2025 (Gmt+09:00)
-
 Business & PoliticsUS tariffs add risk premium to dollar assets: Maurice Obstfeld
Business & PoliticsUS tariffs add risk premium to dollar assets: Maurice ObstfeldApr 29, 2025 (Gmt+09:00)
-

Comment 0
LOG IN


